A Bit of Context
My final project for the MA Conservation at Camberwell College of Arts was the conservation of a nineteenth-century watercolour depicting Elphinstone College in Mumbai, by the British architect James Trubshawe (Figs. 1 & 2). The grand building of the college was designed in the Neo-Victorian Gothic style and recently has been named a UNESCO World Heritage Site, within an ensemble of buildings in the same architectural style in Mumbai.
The watercolour is a recent acquisition of the Royal Institute of British Architects (RIBA), having been bequeathed by Trubshawe’s descendants. This project aimed to repair the mechanical damage to ensure the watercolour’s physical integrity and accessibility while reducing the factors which detracted from the aesthetic value and appreciation of the painting.
To Begin With
The watercolour was oversized, most likely a double elephant drawing paper from which the deckle edge had been cut away. The watermark ‘J. Whatman 1864’ was visible along the top left edge of the paper. The watercolour was adhered to a larger backing paper and mounted on canvas, which was wrapped around a strainer, all of which together, measured approximately 1130mm x 830mm. The strainer had three losses, along the top and right outer member and was slightly warped. The canvas had stretched, cockled and become detached from the backing paper in areas (Fig. 3) and the edges had torn away from the strainer along the top right corner. The backing paper had some losses, was acidic, mouldy and foxed and had delaminated in the top left corner (Fig. 4). The primary support had severe, dark tidelines and large tears, with one spanning 400mm.
Salts on the Surface
Efflorescence was visible where white pigment was used to paint a group of figures, highlights in the building columns and a flower in front of the building. They could also be seen in the opaque green watercolour used in a small group of leaves in the foreground. Most of the white pigment had gone, with only a few remaining flakes and smaller pigment particles to be seen (Fig. 5).
The watercolour was examined under ultraviolet (UV) light. Several areas of white pigment fluoresced yellow-green, indicating the pigment zinc oxide (Fig. 6). However, not much could be seen in the areas with the most efflorescence, such as the group of figures, likely due to the change of the zinc oxide to the salt zinc sulphate, which does not fluoresce.
There is a mixture of large and smaller salt crystals within the areas of white pigment, whereas only smaller crystals were observed within the opaque green leaves. Where salts have ruptured through the paper, some were large enough to cause pits in the paper (Fig. 7).
Considerations to Fix
The washing of the watercolour was complex for several reasons: the large tear, dark tidelines, gum arabic glazes, the efflorescence which was water soluble and the watercolour’s large dimensions. The efflorescence helped to create the colour and forms of the images, where the white pigment had nearly all disappeared. It also provided information such as where a white pigment and a different green watercolour were used. As the painting would be housed at the RIBA in climate-controlled storage and the acidic backing would be removed, the risk of new salt formation was reduced. Therefore, it was decided to keep the efflorescence. The white pigments, glazes and salts needed a temporary fixative during washing.
Molten cyclododecane was tested as a fixative, however the thick barrier was unlikely to have sublimed in time for the painting to be displayed at the Camberwell Summer Show. It was then tested in saturated solutions with “greener” solvents, such as dimethyl carbonate, diethyl carbonate and hexamethyldisiloxne, which allowed for easier application of thinner barriers which could sublime quicker. These barriers were prone to cracking, therefore a layer of Paraloid B72 would have been required. Because of this, it was decided to fix the white pigment and efflorescence with the Paraloid B72 only at 10%, with a couple of layers. Samples of white chalk on paper were used to practice the application and removal of the Paraloid B72 with a rigger brush, without dislodging any chalk particulates. Raking light and transmitted light were used to help see whether areas of efflorescence had been completely protected by the Paraloid B72 (Fig. 8).
For the gum arabic glazes, a less interventive approach was taken. While initial testing showed the glazes were not soluble, due to the prolonged exposure needed for the stain removal, the glazes could begin to swell and drop into the paper. Decamethylcyclopentasiloxane (D5 or cyclomethicone) was chosen as it could be easily brushed on to create a barrier and would slowly evaporate.
Efficiency through Conductivity
The conductivity of the paper and tidelines was tested using a LAQUAtwin-EC-11 compact conductivity meter to analyse 5% agarose gel pellets. By measuring this and adjusting the conductivity of the washing water correspondingly, the washing process was made more efficient, enabling free acids to be drawn out from inside the fibres. This can also be used to manage the swelling of paper fibres during washing (Keynan & Hughes, 2013).
The conductivity of the washing water was set to approximately 5,000μS/cm, which was slightly higher than the measurement of the paper but lower than the tideline. This was achieved by adding acetic acid to deionised water with a further addition of ammonium hydroxide to create a soluble salt solution. This would improve the washing efficiency without causing excess swelling. As a large quantity of adjusted washing water was needed, two batches of the salt solution were made (Figs. 9 & 10).
The Treatment Process
Prior to treatment, the solubility of the pigments was tested and a mock-up of the watercolour was made with the same dimensions and damage. This allowed the equipment to be tested and set up to the correct proportions, as the washing bath had to be made from a sheet of polythene with cedar wood chamber corners. The handling of the object was practised as several people were needed and each needed to know their role. This also allowed the room layout to be checked, to ensure there was enough empty space and tables to place the watercolour, for any emergency.
The canvas was cut away from the strainer and separated from the backing paper with a spatula. The backing paper was removed layer by layer with a scalpel before a methyl cellulose poultice was used to remove the final layer of paper and adhesive. A bleaching effect was seen in relation to the zinc white pigment, and these areas were difficult to wet during the backing removal.
The painting was supported by Bondina® (non-woven support) and humidified using a Gore-Tex© chamber. It was then moved using the Bondina® on top of blotters and sprayed with water to completely wet it before the immersion washing. The blotter absorbed much of the degradation products from the tideline. This resulted in less handling of the watercolour, as there was no need to change the washing water. A sheet of Melinex® was then wetted and the watercolour with the Bondina® support placed on top. The layer of water allowed the painting to sit flat and adhere to the Melinex® due to the surface tension. This protected the large tear.
The watercolour was placed into the washing bath at a low angle, keeping it as flat as possible (Fig. 11). The water was slightly agitated to aid the release of the discolouration trapped underneath the watercolour. The watercolour was moved onto a suction table, which was placed at its lowest height for easier handling, where localised washing of the largest tideline with hot water was attempted (Fig. 12). As the process removed only minimal staining it was not continued. The painting was allowed to air dry.
Further localised cleaning using rigid gels with D5 were tested with no success. Bleaching was also considered but deemed unnecessary, as the tidelines were in the sky and were not detracting from the painting. The removal of the Paraloid B72 was quite slow; however, a pre-treatment with a diethyl carbonate chamber prior to application of the solvent on the suction table sped up the removal process.
The Final Result
The result of washing was the reduction of tidelines and an overall much brighter appearance of the watercolour (Figs. 13-17). The project was a great learning tool, showing the importance of planning and how a helping hand is needed when treating large objects. My colleagues’ support was invaluable for moving and handling the painting. Thank you to the staff and students at Camberwell College of Arts and Joanna Kosek for all their help!

Fig. 13: Detail of tideline and differential darkening due to window mount (before conservation) 
Fig. 14: Tideline reduction and overall brightening (after conservation) 
Fig. 15: Accumulation of degradation along watercolour edge following the window mount, under UV light (before conservation) 
Fig. 16: Reduction in degradation products along watercolour edge, under UV light (after conservation)
Images reproduced with the permission of RIBA Collections.
Reference List:
Keynan, D. and Hughes, A. (2013) Testing the Waters: New Technical Applications for the Cleaning of Acrylic Paint Films and Paper Supports. The Book and Paper Group Annual, 32 43-51. Available from https://cool.conservation-us.org/coolaic/sg/bpg/annual/v32/bp32-08.pdf
Measday, D., Walker, C. and Pemberton, B. (2017) A summary of ultra-violet fluorescent materials relevant to Conservation. Available from https://aiccm.org.au/national-news/summary-ultra-violet-fluorescent-materials-relevant-conservation
Sandbakken, E. S. and Tveit, E. S. (2012) Preserving a Master: Edvard Munch and his painted sketches. Journal of Urban Culture Research, 5 88-104.
Singer, B. and Liddie, C. (2005) A study of unusual degradation on a seascape painting associated with the use of zinc white pigment, The Paper Conservator, 29(1) 5-14.
Stavroudis, C. (2018) Measuring pH and Conductivity with Horiba Meters Available from https://youtu.be/3MkZBxRJ9Yg
Kiri Douglas is a graduate of the BA (Hons) Conservation of Cultural Heritage at Lincoln University (2017), and a graduate of the MA Conservation (Artwork on Paper) at Camberwell College of Arts, London (2019). Having had placements in national institutions and private studios during her studies, Kiri has recently completed a graduate internship in Conservation and Preservation at the Getty Research Institute in Los Angeles, California.






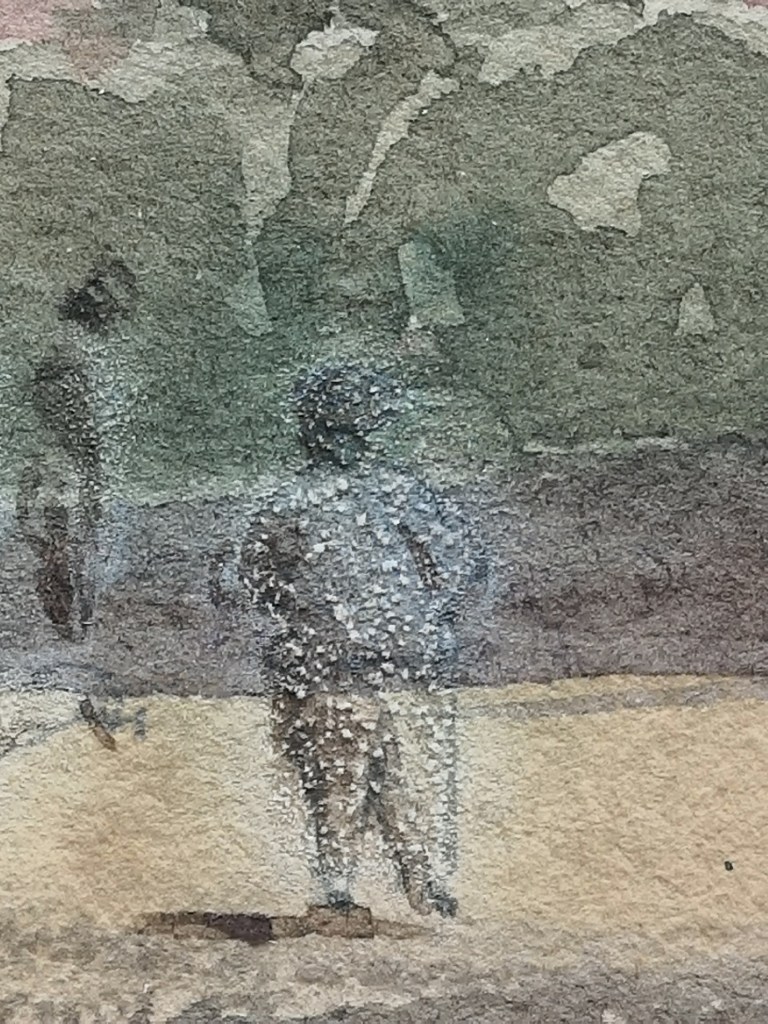






Nice project! Thanks for sharing all these efforts in detail! 🙂
LikeLike
Very interesting project. Lovely to see a project that covers measuring the conductivity of water, which is still a relatively new concept to some.
LikeLike